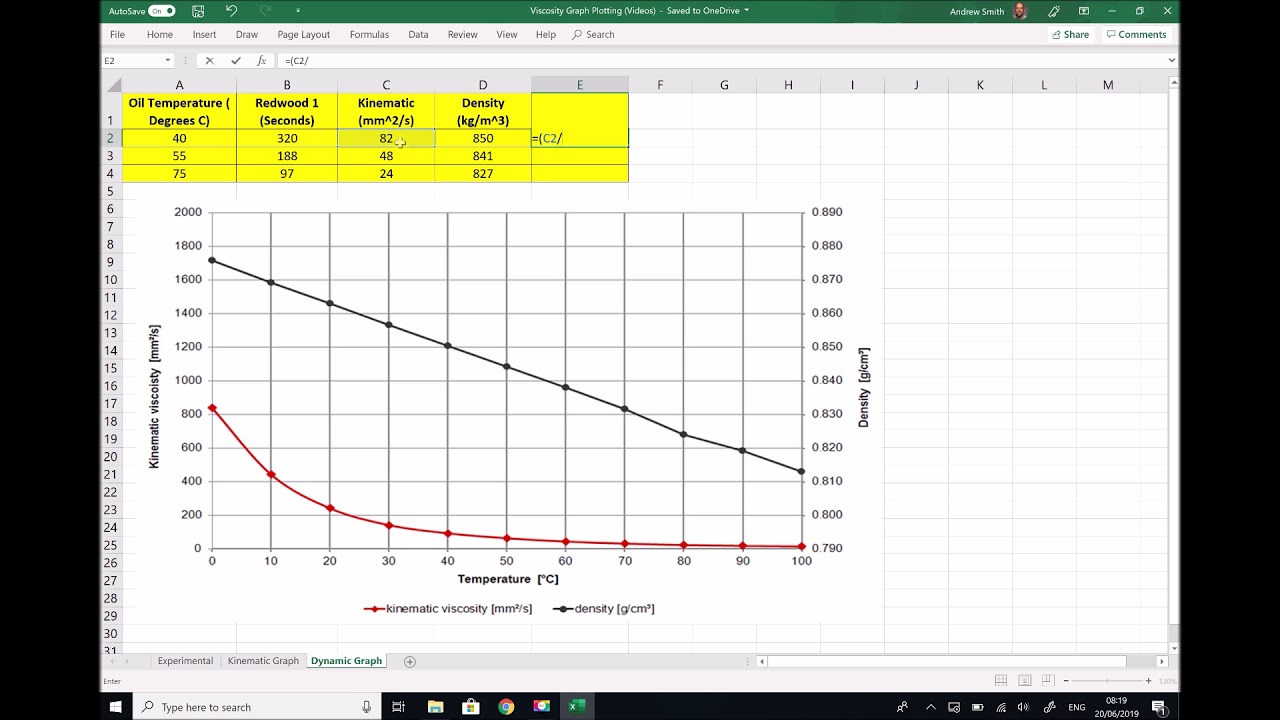
The triple point of a substance is the temperature and pressure at which the three phases (gas, liquid, and solid) of that substance coexist in thermodynamic equilibrium.ĭownload and print Air - Density vs. The curve between the triple point and the critical point shows the air boiling point with changes in pressure.Īt the critical point there is no change of state when pressure is increased or if heat is added.  
The phase diagram for air shows the phase behavior with changes in temperature and pressure. However, at low temperature and high pressures the gas mixture becomes a liquid. See also more about atmospheric pressure, and STP - Standard Temperature and Pressure & NTP - Normal Temperature and Pressure,Īs well as Thermophysical properties of: Acetone, Acetylene, Ammonia, Argon, Benzene, Butane, Carbon dioxide, Carbon monoxide, Ethane, Ethanol, Ethylene, Helium, Hydrogen, Hydrogen sulfide, Methane, Methanol, Nitrogen, Oxygen, Pentane, Propane, Toluene, Water and Heavy water, D 2O.Īir is a mixture of gases at standard conditions. Specific heat (heat capacity) at varying temperature.Specific heat (heat capacity) at varying pressure.Properties at gas-liquid equilibrium condition.Diffusion coefficients of gases in excess of air.Density, specific weight and thermal expansion coefficient at varying temperature.Viscosity, kinematic, at 0☌ and 1 bara: 0.00001349 m 2/s = 13.49 cSt = 0.0001452 ft 2/sįollow the links below to get values for the listed properties of air at varying pressure and temperature:.Specific heat capacity (C v) air at 0☌ and 1 bara: 0.7171 kJ/kgK = 0.17128 Btu(IT)/(lb m ☏) or kcal/(kg K).Specific heat capacity (C p) air at 0☌ and 1 bara: 1.006 kJ/kgK = 0.24028 Btu(IT)/(lb m ☏) or kcal/(kg K).Liquid density at boiling point and 1 bar: 875.50 kg/m 3 = 54.656 lb/ft 3.Bulk modulus elasticity: 1.01325 x 10 5 Pa or N/m 2.Flow of fluids through valves, fittings, and pipe. USA.įor Sutherland's formula and values for : Crane Company. Since Sutherland's formula is an empirical fit of measured data, the following table of reference data is needed.Ī gas with reference viscosity μ 0 = centiPoise,įor gas viscosity: Chemical Rubber Company (CRC). Gas viscosity can be modeled by Sutherland's formula: 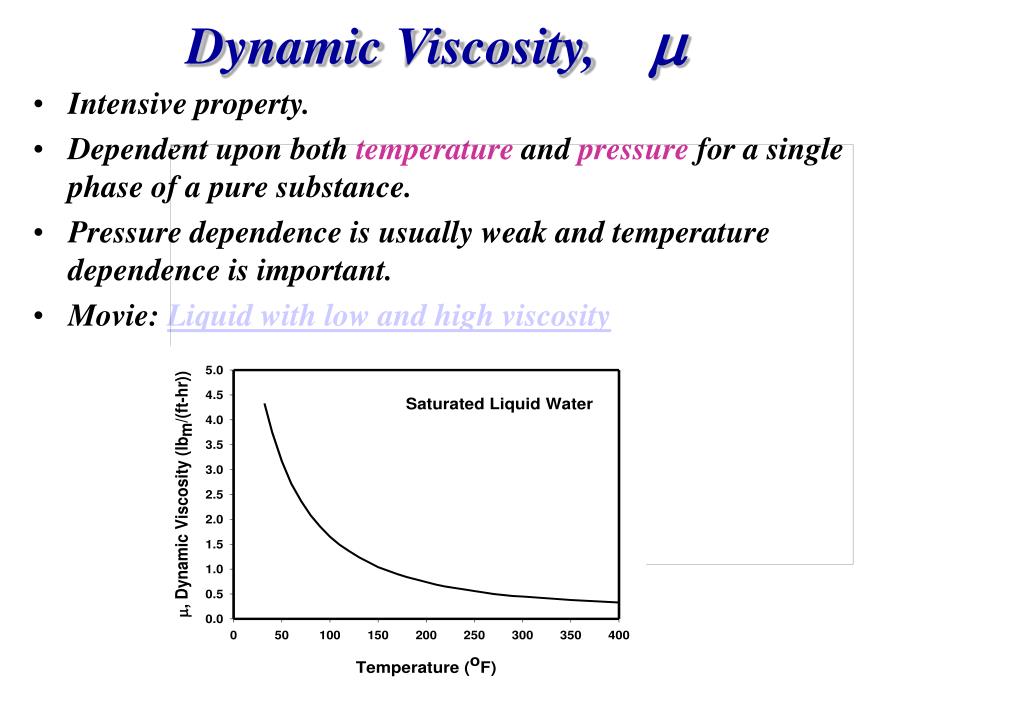
Note that as an engineering quantity, the temperatures used are in the Rankine scale. It is primarily a function of temperature, and can be modeled in terms of temperature with the input of experimental reference measurements. Gas viscosity is only weakly dependent on pressure near atmospheric pressure. The viscosity of gases near room temperature are in the centiPoise range, so that is a commonly used unit. The viscosity of a gas can be thought of as a measure of its resistance to flow and is measured in the CGS unit Poise = dyne sec/cm 2.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
